What Is Biuret Test?
Biuret test is a chemical test used for detecting the presence of peptide bonds in a given analyte. Biuret test is also referred to as Piotrowski’s test, the name of Gustaw Piotrowski, a polish physiologist who first devised and explained the test in 1857.
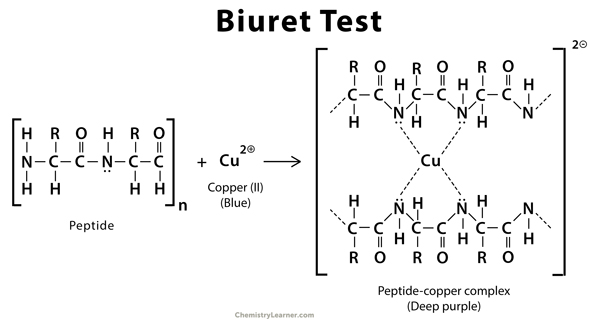
In presence of peptide bonds, a copper II ion forms mauve-colored complexes (pale-purple) in an alkaline solution. The biuret test can be used to assess the concentration of proteins because peptide bonds occur with the same frequency per amino acid in the peptide. The intensity of the color and hence the absorption at 540 nm, is directly proportional to the protein concentration.
Objective Of Biuret Test
- To detect the protein in the given solution.
- To analyze the presence of the peptide bond in a given analyte.
Biuret Reagent
The Biuret reagent is a solution composed of sodium hydroxide (NaOH) or potassium hydroxide (KOH), hydrated copper (II) sulfate, and potassium sodium tartrate. Sodium hydroxide and Potassium hydroxide provide the alkaline medium and potassium sodium tartrate is added to chelate and thus stabilize the cupric ions in the solution or to maintain their solubility in alkaline solution. The reaction of the cupric ions with the nitrogen atoms involved in peptide bonds leads to the displacement of the peptide hydrogen atoms under the alkaline conditions.
Principle Of Biuret Test
The biuret method is a colorimetric technique specific for proteins and peptides. Copper salts in alkaline solution form a purple complex with substances containing two or more peptide bonds. In other words, Biuret test is based on the reaction of the cupric ions Cu2+ with peptide bonds in an alkaline solution. These ions react with the nitrogen of the peptide bond to form a purple or violet colored complex. In alkaline medium, cupric hydroxide is generated from copper sulfate of biuret reagent that helps in chelating the peptide bond with cupric ions to give violet or purple color. This chelate complex has the ability to absorb light with a wavelength of 540nm, which imparts a purple color to it. Therefore, the formation of a purple coloured complex indicates the presence of proteins in the analyte. The absorbance produced is proportional to the number of peptide bonds that are reacting and therefore to the number of protein molecules present in the reaction system. Short-chain peptides often yield blue or pink color in the biuret test.
Experiment
Reagent And Material Required
- 1 % alanine, 5 % egg white (albumin)
- Biuret reagent
- Deionized water
- Water bath
- Dry test tubes
- Pipettes
Biuret Test Procedure
- Take 3 clean and dry test tubes.
- Add 1-2 ml of the test solution, egg albumin, and deionized water in the respective test tubes.
- Add 1-2 ml of Biuret reagent to all the test tubes.
- Shake well and allow the mixtures to stand for 5 minutes.
- Observe for any color change.
Biuret Test Result Interpretation
- Positive Test: A positive biuret test is indicated by presence of blue to deep purple color in the test tube. This confirms presence of peptide bonds.
- Negative Test: A negative biuret test is indicated by absence of purple color in the test tube. This confirms absence of peptide bonds.
Uses Of Biuret Test
- It can be used to detect the amount of protein in the urine.
- Biuret reaction with protein is applicable to the quantitative determination of total protein by spectrophotometric analysis.