Molecules of organic compounds except that of hydrocarbons can be divided into two parts, a reactive part which is referred to as functional group and a skeleton of carbon atoms called alkyl group. The properties of a compound are largely determined by the functional group. Different compounds having same functional group have similar properties and are classified as family of compounds. Compounds having different functional groups have different properties and belong to different families of compounds.
Some of the common functional groups present in organic compounds are:
Saturated And Unsaturated Hydrocarbons
Hydrocarbons, or molecules which contain hydrogen atoms and carbon atoms that are bonded to each other and may or may not contain other atoms, are classified into two distinct categories: saturated and unsaturated.
Saturated Hydrocarbons
A Saturated hydrocarbon is a hydrocarbon in which all the carbon-carbon bonds are single bonds. A hydrocarbon is an organic compound whose only constituents are carbon and hydrogen. As the name suggests, saturated hydrocarbons are hydrocarbons in which all the carbon atoms are bonded to four other atoms and are ‘saturated’, implying that no carbon-carbon multiple bonds exist in these organic compounds.Generally, the term ‘saturated hydrocarbon’ is used to refer to alkanes – acyclic hydrocarbons containing only sp3 hybridized carbon atoms.
A few other examples of saturated hydrocarbons include:
- Butane (C4H10)
- Octane (C8H18)
- Cyclohexane (C6H12)
- Cyclopropane (C3H6)
Unsaturated Hydrocarbons
Unsaturated hydrocarbons are organic compounds that are entirely made up of carbon and hydrogen atoms and consist of a double or a triple bond between two adjacent carbon atoms. The hydrocarbons having at least one double bond between two adjacent carbon atoms are called alkenes, whereas the hydrocarbons which contain a carbon-carbon triple bond are referred to as alkynes.
Examples Of Unsaturated Hydrocarbons
- Ethene, C2H4
- Benzene, C6H6
- Acetic acid, C2H4O2
Difference Between Saturated and Unsaturated Hydrocarbon
Unsaturated hydrocarbons feature at least one double or triple bond between two adjacent carbon atoms. The key differences between saturated and unsaturated hydrocarbons are tabulated below.
| Saturated Hydrocarbon | Unsaturated Hydrocarbon |
| All carbon atoms are sp3 hybridized in these compounds. | They contain sp2 or sp hybridized carbons. |
| Contain more hydrogen atoms than the corresponding unsaturated hydrocarbons. | Contain fewer hydrogens than the corresponding saturated hydrocarbon. |
| Examples include alkanes and cycloalkanes. | Examples include alkenes, alkynes, and aromatic hydrocarbons. |
| They have a relatively low chemical reactivity | They are more reactive than their saturated counterparts. |
| They generally burn with a blue flame | They generally burn with a sooty flame. |
Tests for Unsaturation
There are two methods for detecting the unsaturation in an organic compound.
- Alkaline potassium permanganate test (Baeyer’s test)
- Bromine test
Materials Required
- Potassium hydroxide solution
- Bromine water solution/ Bromine in CCl4 or chloroform
- Carbon tetrachloride/chloroform
- Potassium permanganate solution
- Sample compound to be tested
- Distilled water
- Test tubes
- Test tube holder
Preparation of Reagents
- Alkaline KMnO4 (1%): Dissolve about 1g of potassium permanganate in 100ml of distilled water. To that solution add 10g of anhydrous sodium carbonate. Shake to dissolve it completely.
- Bromine in CCl4: Take 5ml of liquid bromine in 100ml of carbon tetrachloride and shake it to dissolve properly.
- Bromine Water: Take 5ml of bromine add 100ml of distilled water and shake well. Decant off the clear liquid.
1. Bromine Test
Bromine solution is brown. In this test when bromine solution is added to the unsaturated hydrocarbon the brown colour disappears if the hydrocarbon is unsaturated. Bromine forms an addition product with the unsaturated hydrocarbon.

Procedure
When the compound is soluble in water.
- Dissolve a part of the given compound (solid or liquid) in about 2 ml of distilled water in a test-tube and add a drop of bromine water and shake.
- Disappearance of orange-red colour of bromine indicates unsaturation. Continue the addition of bromine water drop wise with constant shaking. The disappearance of orange- red colour continues so long as there are unsaturated bonds. When all the pi-bonds are broken, the orange-red colour shall persist. Bring a rod dipped in NH4OH near the mouth of the test-tube.
- Absence of white fumes confirms unsaturation.
When the compound is insoluble in water.
- Dissolve a small amount of the given compound in 1 ml of CCl4 in a test tube and add 2% solution of bromine in CCl4. Shake the mixture.
- Disappearance of orange-red colour indicates unsaturation.
- Continue the drop wise addition of Br2 with constant shaking until the brown colour persists. Bring a rod dipped in NH4OH near the mouth of the test-tube. Absence of white fumes confirms unsaturation.
When the compound is a gas.
- Add 1-2 ml of bromine solution into a gaseous compound taken in a jar and shake. Disappearance of orange-red colour of bromine indicates unsaturation.
- Note. No HBr is evolved in bromine test for unsaturation. However, some compounds such as aniline form substituted products with bromine and evolve HBr.
- Caution: Handle bromine solution carefully. Avoid its context with skin.
2. Alkaline KMnO, Test (Baeyer’s Test)
In this test the pink colour potassium permanganate disappears when an alkaline potassium permanganate is added to an unsaturated hydrocarbon. The disappearance of pink colour may take place with or without the formation of brown precipitate of manganese oxide.

Procedure
- Take a little of the given organic compound to be tested in a test tube.
- Dissolve the organic compound in 2ml of water or acetone in the test tube.
- Add 1% alkaline potassium permanganate solution dropwise and shake the mixture.
- Observe the solution, if pink colour persists then it is saturated compound. If the pink colour disappears then the given organic compound is unsaturated.
Tests for the Alcoholic Group [R-OH]
Alcohols
Compounds in which the hydroxyl group (—OH) is linked to aliphatic carbon chain or in the side chain of an organic compound are called alcohols. The alcohols containing one, two or three hydroxyl groups per molecule are called mono, di or trihydric alcohols respectively.
Alcohols are further classified as primary (1°), secondary (2°) and tertiary (3°) according as the —OH group is attached to primary, secondary and tertiary carbon atoms respectively.

The alcoholic group can be detected by any of the following tests:
1. Sodium metal test.
2. Ester test.
3. Ceric ammonium nitrate test.
4. Acetyl chloride test.
5. Iodoform test for alcohols containing the

Materials Required
- Sodium metal
- Acetic acid
- Ceric ammonium nitrate
- Acetyl chloride
- Ammonium hydroxide
- Iodine
- Sodium hydroxide
- Calcium sulfate
- Lucas reagent (zinc chloride and Con.HCl)
- Test tubes
- Test tube holder
- Filler
Preparation of Reagents:
- Ceric Ammonium Nitrate: Take 20gm of orange crystals of ceric ammonium nitrate in 200ml of warm dilute nitric acid.
- Lucas Reagent: Take 135gm of anhydrous zinc chloride and dissolved in 100ml of concentrated hydrochloric acid.
- Iodine Solution: Dissolve 5gm of potassium iodide in 40ml of water. To that solution add 1gm of solid iodine and dissolve it completely.
- 5% sodium hydroxide solution. Dissolve 5 g of solid sodium hydroxide in about 50 ml water and then make up the volume to 100 ml.
Sodium metal test
This test is based on the appearance of effervescence due to liberation of hydrogen gas when the alcohol is reacted with active metals like sodium.

The alcohol to be tested should be dry because water also reacts with sodium. Sodium should be handled carefully, unreacted sodium should be destroyed by adding excess alcohol. This test is favourable if phenyl or carboxyl groups are absent.
Procedure
- Take the organic compound to be tested in a dry test tube.
- Add 1gm of anhydrous calcium sulfate and shake well to remove excess water.
- Decant the solution to another clean test tube.
- Add a small piece of sodium metal.
Results: If brisk effervescence appears due to the evolution of hydrogen gas indicate the presence of alcoholic group.
Ester test
Alcohols react with carboxylic acids to form sweet smelling esters. The reaction between an alcoholic group and a carboxylic acid group is called esterification. This is a slow and reversible reaction and is catalysed by an acid such as cone, sulphuric acid.
The chemical reaction is given below:

Procedure
- Take 1ml of the organic liquid to be tested in a clean dry test tube.
- Add 1ml of glacial acetic acid and 2-3 drops of conc. sulfuric acid.
- Heat the mixture in a water bath for 10 minutes.
- The hot mixture is poured into a beaker containing cold water.
- Smell the water in the beaker.
- A fruity smell confirms the presence of alcoholic group.
Ceric ammonium nitrate test
Alcohol or reaction with ceric ammonium nitrate forms a pink or red colour precipitate due to the formation of a complex compound and ammonium nitrate.
Here is the chemical reaction

Procedure
- Take 1ml of given compound in a dry test tube.
- Add a few drops of ceric ammonium nitrate reagent and shake the solution well.
- Observe the solution.
- If red precipitate appears then the presence of alcoholic group is conformed.
Results: The appearance of wine red colour precipitate shows the presence of alcoholic group.
Acetyl chloride test
Alcohol reacts with acetyl chloride results in the formation of ester and hydrogen chloride. The resulting hydrogen chloride on contact with ammonium hydroxide forms a white fumes of ammonium chloride and water.

Procedure
- Take 2ml of given organic compound in a clean test tube
- Add 1gm of anhydrous calcium sulfate and shake well.
- Filter the solution. To the filtrate add 3 to 4 drops of acetyl chloride and shake well.
- Take a glass rod dipped in ammonium hydroxide solution.
- Bring the glass rod near the mouth of the test tube.
Results: The formation of white fumes indicates the presence of alcohol.
Iodoform test
This test is given by secondary alcohols, ketones and acetaldehyde. First the compound is heated with sodium hydroxide solution and iodine. A formation of yellow precipitate of iodoform shows the presence of alcohol.

Procedure
- Take 1ml of given organic compound in a clean dry test tube.
- Add 1ml of 1% iodine solution to it.
- Add dilute sodium hydroxide solution drop wise until the brown colour of iodine is discharged.
- Heat the mixture gently in water bath.
Results: The formation of yellow precipitate shows the presence of alcohol, acetaldehyde or methyl ketones.
Distinction Between Primary, Secondary and Tertiary Alcohols
Lucas Test
This test is based upon relative reactivities of various alcohols towards HCl in the presence of ZnCl2. In this test, the alcohol is treated with Lucas reagent which is equimolar mixture of HCl and ZnCl2. The reagent It reacts with primary, secondary and tertiary alcohols at different rates. This reagent forms a cloudiness on reacting with alcohols. Tertiary alcohols reacts immediately and give cloudiness, secondary alcohols reacts slowly and gives cloudiness after 5 to 10 minutes and there is no reaction with primary alcohols.

Procedure
- Take about 1 ml of dry alcohol in a clean dry test-tube and add 8-10 drops of Lucas reagent.
- Shake the mixture well.
- Note down the appearance of cloudiness.
Result Interpretation
- If cloudiness appears immediately, tertiary alcohol is indicated.
- If cloudiness appears within five minutes, secondary alcohol is indicated.
- If cloudiness appears only upon heating, primary alcohol is indicated.
Tests for Phenolic Group [Ar—OH]
Phenols
Phenol (also called carbolic acid) is an aromatic organic compound with the molecular formula C6H5OH. It is a white crystalline solid that is volatile. The molecule consists of a phenyl group (−C6H5) bonded to a hydroxy group (−OH). Mildly acidic, it requires careful handling because it can cause chemical burns.
Phenol was first extracted from coal tar, but today is produced on a large scale (about 7 billion kg/year) from petroleum-derived feedstocks. It is an important industrial commodity as a precursor to many materials and useful compounds. It is primarily used to synthesize plastics and related materials. Phenol and its chemical derivatives are essential for production of polycarbonates, epoxies, Bakelite, nylon, detergents, herbicides such as phenoxy herbicides, and numerous pharmaceutical drugs.

Tests for Phenolic [Ar—OH] Group
The phenolic group can be detected by the following tests:
- Litmus test
- Ferric chloride test
- Libermann’s test
- Bromine water test
- Phthalein dye test
Materials Required
- Blue litmus paper
- Ferric chloride solution
- Sodium nitrite
- Concentrated sulfuric acid
- Sodium hydroxide
- Bromine water
- Phthalic anhydride
- Organic compound to be tested
- Test tubes
- Test tube holder
- Dropper
- Beaker
Litmus Test
In laboratory, a litmus paper is used to test whether the given solution is acidic or basic. Red litmus paper turns blue while blue litmus paper remains unchanged in the presence of a base.
Procedure
- Place the drop of given organic solution or a small crystal on moist blue litmus paper.
- Observe the change in colour, if it changes to red then phenolic group may be present.
Phenol turns blue litmus paper red. This shows that phenol is acidic in nature. Carboxylic acid also give this test. Compare to carboxylic acid phenol is weakly acidic and it does not give an effervescence with aqueous sodium carbonate.
Ferric Chloride Test
Aqueous solution of phenol reacts with freshly prepared ferric chloride solution gives coloured complex. Most phenols give dark coloured solution.Ferric chloride solution: Neutral solution of ferric chloride is prepared by adding diluted solution of sodium hydroxide to ferric chloride solution drop by drop until a small but permanent brown precipitate appears. Filter the solution and use the clear filtrate for the test.
Here is the chemical reaction

Procedure
- Dissolve the given organic compounds in water.
- Add neutral solution of ferric chloride slowly dropwise.
- Observe the change in colour.
- A red, blue, green or purple colouration indicates the presence of phenol.
Note:
1.o, m, p-cresol, resorcinol give violet or blue colouration.
2. β-Naphthol gives a green colouration.
3. α-Naphthol gives pink colouration.
4. Formic acid and acetic acid give deep red colouration.
Liebermann’s Test
Phenol reacts with concentrated sulfuric acid and sodium nitrite forms a yellow colour quinone monoxime complex. With excess of phenol and sulfuric acid a deep blue indophenol complex is formed. On dilution a red colour indophenol is formed which turns to deep blue colour sodium salt solution of indophenol on treatment with sodium hydroxide.


Procedure
- Place the crystals of sodium nitrite in a clean dry test tube.
- Add 1ml of phenol to sodium nitrite solution.
- Heat the mixture gently and allow it to cool.
- Add 1ml of concentrated sulfuric acid to it and shake the contents.
- Observe the change in the colour of the solution.
- Dilute the solution with water so that the given compound turns red if phenolic group is present.
- Now add sodium hydroxide solution, the blue colour solution or green colour solution appears.
Note:
1. Nitrophenols and p-substituted phenols do not give this test.
2. Among the dihydroxyphenols, only resorcinol gives positive test.
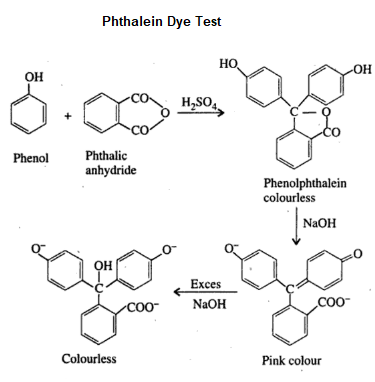
Phthalein Dye Test
Phenol on heating with phthalic anhydride in the presence of concentrated sulfuric acid forms a colourless condensation compound called phenolphthalein. On further reaction with dilute sodium hydroxide solution gives a pink colour fluorescent compound called fluorescein. Characteristic colours are produced by different phenolic compounds which can be viewed under white background.
Here is the chemical reaction:
Procedure
- Take the organic compound to be tested in a test tube.
- Add 200mg of phthalic anhydride to it.
- Add drops of concentrated sulfuric acid to the mixture.
- Heat the solution for 2-3 minutes.
- Cool the mixture and pour it into a beaker containing dilute sodium hydroxide solution.
- Dilute the whole mixture with equal volume of water.
- Observe the change in the colour in a white background.
- If fluorescence colour exists the view it in a black background.
The colours produced by different phenolic compounds in phthalein dye test is given below
| Phenol | Reddish pink |
| o-cresol | Red |
| m-cresol | blue or violet blue |
| 1-naphthol | green |
| 2-naphthol | faint green |
| Resorcinol | yellow-green fluorescence |
| Hydroquinone | deep purple |
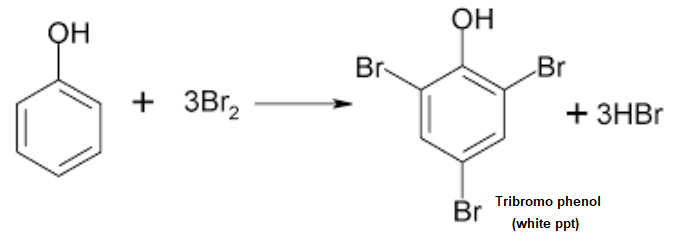
Bromine Water Test
Phenol undergoes electrophilic substitution reaction with bromine. When bromine water is added to aqueous solution of phenol the brown colour of bromine disappears and a white precipitate of tribromophenol is formed.
Here is the chemical reaction
Procedure
- Take 5ml of bromine add 100ml of distilled water and shake well. Decant off the clear liquid.
- Dissolve the given organic compound in glacial acetic acid.
- Add bromine water solution to this dropwise.
- If the colour of bromine disappears then it indicates the presence of phenol.
Uses Of Phenols
- Phenol is used as an antiseptic and disincentive agent.
- Phenol has been used in many cosmetics preparations such as sun screens, hair colorings and skin formulations.
- Longer alcohols and fatty alcohols have been used in the industries of plasticizers and detergents.
- Some phenolic compounds are used in food additives.
- Phenolic compounds are used as drugs and prodrugs for treatments of several diseases such as Parkinson’s disease.
- Some phenols are used in chemical synthesis procedures or as indicators.
Tests for Aldehydic and Ketonic [—CHO and —CO—] Groups
Aldehydes and Ketones
Aldehydes and ketones have a carbonyl group (C=O) as a functional group. A ketone has two alkyl or aryl groups attached to the carbonyl carbon (RCOR’). The simplest ketone is acetone, which has two methyl groups attached to the carbonyl carbon (CH3COCH3).
An aldehyde is similar to a ketone, except that instead of two side groups connected to the carbonyl carbon, they have at least one hydrogen (RCOH). The simplest aldehyde is formaldehyde (HCOH), as it has two hydrogens connected to the carbonyl group. All other aldehydes have one hydrogen bonded to the carbonyl group, like the simple molecule acetaldehyde, which has one hydrogen and one methyl group (HCOCH3).
The carbonyl carbon in both aldehydes and ketones is electrophilic, meaning that it has a dipole due to the electronegativity of the attached oxygen atom. This makes the carbonyl carbon an ideal target for nucleophiles in a nucleophilic addition reaction. During this reaction, the nucleophile, or electron donor, attacks the carbonyl to form the tetrahedral intermediate. The negatively charged oxygen accepts a hydrogen ion to form a hydroxyl group.
Typically, nucleophiles possess a negative charge or lone pair on a heteroatom, which can take several forms (OH–, RO–, CN–, R3C–, RNH2, ROH). For primary amines (RNH2), the reaction does not stop at the formation of the tetrahedral intermediate with a hydroxyl group. Rather, an elimination reaction occurs that produces a double-bonded carbon and nitrogen functional group known as an imine. Understanding the reactions that aldehydes and ketones can undergo provides a way to differentiate between these similar organic compound types.
This group has characteristic properties which are shown by both classes of compounds. In aldehydes, the carbonyl group is attached to a hydrogen atom and to an aliphatic or aromatic radical. Formaldehyde is an exception in which the carbonyl group is attached to two hydrogen atoms.

The carbonyl group in aldehydes and ketones is identified by the following tests:
1. 2, 4-dinitrophenyl hydrazine test.
2. Sodium bisulphite test.
The difference between ketone and aldehyde is the carbonyl group present in aldehydes can be easily oxidised to carboxylic acids whereas the carbonyl group in ketones are not oxidised easily. This difference in reactivity is the basis for the distinction of aldehydes and ketones. They are generally distinguished by the following tests.
- Schiff’s test
- Fehling’s test
- Tollen’s test
- Test with chromic acid
- Sodium nitroprusside test
Materials Required
- Schiff’s reagent
- Silver nitrate solution
- Fehling’s solutions A
- Fehling’s solutions B
- Dilute ammonium hydroxide solution
- 2,4-Dinitrophenylhydrazine reagent
- Chromic acid
- Sulfuric acid
- Sodium bisulfite
- Test tubes
- Test tube holder
- Beaker
2, 4-Dinitrophenyl hydrazine Test (2, 4—DNP Test)
Procedure
- Dissolve the given organic compound in ethanol.
- To this solution add alcoholic reagent of 2,4-dinitrophenyl hydrazine.
- Shake the mixture well.
- If there is a formation of yellow to orange precipitate then the given compound is an aldehyde or ketone.
- An orange precipitate is obtained from carbonyl compounds in which the C=O groups is conjugated with C=C.

Note: Formation of orange-yellow crystals indicates the presence of carbonyl group.
Sodium bisulphite Test
Aldehydes and ketones combine with sodium bisulfite to for well-crystallized water-soluble products known as “aldehyde bisulfite” and “ketone bisulfite”.
Procedure
- Take a saturated solution of sodium bisulfite in a clean test tube.
- Add 1ml of the given organic compound to be tested.
- Shake well and leave it for 15-20 minutes.
- If there is a formation of white precipitate then the presence of the carbonyl group is confirmed.
The chemical reaction

Formation of crystalline precipitate confirms carbonyl group
Note
- Formaldehyde, acetaldehyde, benzaldehyde, acetone, methyl ethyl ketone and diethyl ketone give position tests.
- Aqueous solutions of formaldehyde and acetaldehyde form addition products but as they are highly soluble , precipitates are rarely formed.
- Acetophenone and benzophenone do not give this test.
Differentiating Tests for Aldehydes
The following tests are given by aldehydes but not by ketones:
Schiff’s Test
Schiff’s reagent is prepared by passing sulfur dioxide into a solution of the dye fuchsin. The solution becomes colourless due to the formation of an additional product. Aldehydes abstract sulfurous acid from the Schiffs reagent and restores the pink colour. The colouration is due to the formation of complex compound. Ketones, in general, do not respond to this reaction.The reaction should not be subjected to heat. Some ketones give a light pink colour with Schiff’s reagent therefore light pink colour formation is not a positive test.
Procedure
- Take the given organic compound to be tested in a clean test tube.
- Add 2-3 drops of Schiff’s reagent.
- If there is instant pink or red colour formation then the presence of aldehyde is confirmed.
Result: Appearance of pink, red or magenta colour indicates the presence of aldehyde group.
Note:
- The Schiffs reagent should not be warmed.
- The Schiff s reagent should not be treated with alkalies. Otherwise the pink colour develops even in the absence of aldehydes.
- With benzaldehyde the pink colour develops slowly.
Tollen’s Test (Silver mirror test)
This test is also called the silver mirror test. Tollens reagent consists of silver ammonia complex in ammonia solution. Aldehydes reacts with Tollens reagent gives a grey-black precipitate or a silver mirror. Always a freshly prepared Tollen’s reagent should be used. Aldehydes are oxidised to the corresponding acid and silver in Tollens reagent is reduced from +1 oxidation state to its elemental form. Generally ketones do not respond to this test.

Apart from aldehydes some other compounds also respond to Tollen’s test, but the presence of aldehydes is conformed when the given substance shows a positive test for Tollens test but if the given compound passes 2,4-dinitrophenylhydrazine test.
Procedure
- Take 1ml of silver nitrate solution in a clean test tube.
- Add dilute sodium hydroxide solution to it, a brown precipitate forms.
- Add dilute ammonia solution dropwise till the brown precipitate of silver oxide dissolves.
- To this freshly prepared Tollen’s reagent add the given organic compound to be tested.
- Place the test tube in a warm water bath for about 5 to 10 minutes.
- If there is the appearance of a silver mirror in the sides of the test tube conforms the presence of an aldehyde.
Results: The appearance of shiny silver mirror confirms the presence of aldehydes.
Note:
- Many other types of compounds give positive silver mirror test but they do not give 2,4-dinitro phenyl hydrazine test.
- Formic acid, tartaric acid and many carbohydrates like glucose give silver mirror test.
Fehling’s Test
Feling’s solution is a complex compound of Cu2+. When aldehyde compound is treated with Fehling’s solution Cu2+ is reduced to Cu+ and the aldehyde is reduced to acids. During the reaction, a red precipitate is formed. Aromatic aldehydes do not respond to Fehling’s test. An aqueous solution of the compound may be used instead of an alcoholic solution. Formic acid also give this test.

Procedure
- Fehling’s solution is prepared by mixing equal amounts of Fehling’s A and Feling’s B solution.
- Take the given organic compound in a clean test tube.
- Add Fehling’s solution to it and heat the solution gently.
- If a brick-red precipitate appears then the presence of aldehyde is conformed.
Result: Appearance of red precipitate confirms the presence of an aldehydic group.
Note:
1. Benzaldehyde may or may not give this test as the reaction is very slow.
2. Formic acid also gives this test.
Test with Chromic Acid
Aldehydes react with chromic acid gives a green to blue precipitate. Ketones do not react with chromic acid. Some of the primary and secondary alcohols also give this test but they do not give dinitrophenylhydrazine test.
R-CHO + 2CrO3 + 3H2SO4 → 3R-C(O)-OH + 3H2O + Cr2(SO4)3(green colour)
Procedure
- Take the given organic compound in a clean test tube.
- Add 1ml of chromic acid reagent to the given organic compound.
- The appearance of a green or blue colour precipitate indicates the presence of aldehydes.
Result: The appearance of green or blue colour precipitate confirms the presence of aldehydes.
Sodium Nitroprusside Test
Ketone responds to this test. Ketone reacts with alkali forms an anion further it reacts with sodium nitroprusside forms a coloured complex ion. Aldehydes do not respond to this test.
Here is the chemical reaction
CH3COCH3 + OH–→ CH3COCH2– + H2O
[Fe(CN)5NO]2- + CH3COCH2– → [Fe(CN)5NO.CH3COCH2]3-
Procedure
- Dissolve sodium nitroprusside in distilled water in a clean test tube.
- Add 1ml of the given organic compound to be tested.
- Shake well and add sodium hydroxide solution dropwise.
- If there is the appearance of red colour then the presence of ketone is conformed.
Result: The appearance of red colouration shows the presence of ketone.
Differentiating Tests for Ketones
The following tests are given by ketones but not by aldehydes:
m-Dinitrobenzene test
Place 0.5 ml of the given liquid (or 0.5 g of solid) in a clean test-tube and add about 0.1 g of finely powdered m-dinitrobenzene. Now add about 1 ml of dilute sodium hydroxide solution and shake.
Appearance of violet colour which slowly fades confirms ketonic group.
Note: Benzophenone does not give this test.
Sodium nitroprusside test
The anion of the ketone formed by an alkali reacts with nitroprusside ion to form coloured ion.

Procedure
Dissolve a crystal of sodium nitroprusside in about 1 ml of distilled water in a clean test-tube and then add 0.5 ml (or 0.5 g) of the given compound. Shake and add sodium hydroxide solution drop wise.
A red colouration indicates the ketonic group.
Note:
1. Benzaldehyde also gives red colour,
2. Benzophenone does not give this test.
Preparation of Reagents
1. 2, 4-Dinitro phenylhydrazine (2, 4-DNP). Dissolve 1 g of 2, 4-dinitrophenyl hydrazine in 50 ml methanol to which 2 ml of concentrated sulphuric acid is added. Filter, if necessary.
2. Sodium bisulphite, NaHSO3. Dissolve about 30 g sodium bisulphite in 100 ml of distilled water.
3. SchifFs reagent. Dissolve 0.1 g p-rosanaline hydrochloride in 100 ml water and pass sulphur dioxide gas until its red colour is discharged. Filter and use the filtrate.
4. Fehling’s solution A. Dissolve 35 g of crystalline copper sulphate in 500 ml water and add 2 ml cone. H2S04
5. Fehling’s solution B. Dissolve 173 g of Rochelle salt (sodium potassium tartrate) and 60 g sodium hydroxide in 500 ml water.
Tests for Carboxylic Group [—COOH]
Carboxylic Acids
Carboxylic acid, any of a class of organic compounds in which a carbon (C) atom is bonded to an oxygen (O) atom by a double bond and to a hydroxyl group (―OH) by a single bond. A fourth bond links the carbon atom to a hydrogen (H) atom or to some other univalent combining group. The carboxyl (COOH) group is so-named because of the carbonyl group (C=O) and hydroxyl group.
The chief chemical characteristic of the carboxylic acids is their acidity. They are generally more acidic than other organic compounds containing hydroxyl groups but are generally weaker than the familiar mineral acids (e.g., hydrochloric acid, HCl, sulfuric acid, H2SO4, etc.).
Carboxylic acids occur widely in nature. The fatty acids are components of glycerides, which in turn are components of fat. Hydroxyl acids, such as lactic acid (found in sour-milk products) and citric acid (found in citrus fruits), and many keto acids are important metabolic products that exist in most living cells. Proteins are made up of amino acids, which also contain carboxyl groups.
Compounds in which the ―OH of the carboxyl group is replaced by certain other groups are called carboxylic acid derivatives, the most important of which are acyl halides, acid anhydrides, esters, and amides.
Carboxylic acids can be identified by the following tests:
- Litmus test.
- Sodium bicarbonate test.
- Ester test.
- Fluorescein test
Materials Required
- Blue litmus paper
- Sodium bicarbonate (or) sodium hydrogen carbonate
- Ethyl alcohol
- Concentrated sulfuric acid
- Resorcinol
- Acid anhydride
- Given organic compound
- Test tubes
- Test tube holders
- Beaker
- Glass rod
- Stirrer
Litmus Test
The carbobxylic acids turn blue litmus red. The hydroxyl group in —COOH is far more acidic than in alcohol.

- Add a drop of given organic compound on blue litmus paper.
- Observe the colour change in blue litmus paper.
- If the colour of blue litmus changes to red the presence of carboxylic acid.
Note: Blue litmus solution is also used in the place of blue litmus paper.If the colour of the blue litmus paper changes to red then carboxylic group is present. Phenol also gives this test.
Sodium Bicarbonate Test
Carboxylic acids react with sodium hydrogen carbonate to give carbon dioxide gas which is identified by the effervescence produced. This test is used to distinguish carboxylic acids from phenols.

- Prepare a saturated solution of sodium bicarbonate by dissolving sodium bicarbonate in 1ml of water.
- Add the given organic compound on the saturated solution of sodium bicarbonate solution.
- Shake the solution well.
- If there is an evolution of brisk effervescence then it indicates the presence of carboxylic acid.
Note: Use acid free alcohol for the test.
Ester Test
A carboxylic acid reacts with an alcohol in presence of a little sulphuric acid to form an ester which is recognized by its fruity smell.

Procedure
- Mix the given compound with ethyl alcohol and concentrated sulfuric acid.
- Heat the mixture in a dry test tube in a water bath.
- Pour the reaction mixture into a beaker carefully containing water.
- Neutralise the excess sulfuric acid.
- If a sweet smelling substance is sensed then it indicates the presence of acid.
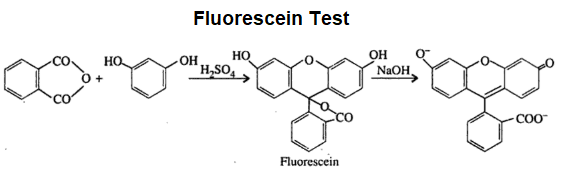
Fluorescein Test
This test is given by dicarboxylic acid. Dicarboxylic acid on heating gives acid anhydride. When this anhydride is treated with resorcinol in the presence of concentrated sulfuric acid a fluorescent
dye is formed and so this reaction is called fluorescein test.
The chemical reaction is given below.
Note: This test should be performed only if the compound gives positive results in litmus test and sodium bicarbonate test.
Tests for Amino Group [—NH3]
Amines
Amines may be considered as substitution products of ammonia. When one hydrogen atom of ammonia is replaced with an alkyl or aryl group,the resulting amine is called primary amine (R—NH2). When two hydrogen atoms of ammonia molecule are replaced with two alkyl or aryl groups, the resulting compound is called secondary amine. The replacement of all the three hydrogen atoms of ammonia with alkyl and aryl groups gives tertiary amine.

There are 5 tests that can be carried out for amino functional group:
- Solubility Test
- Litmus Test
- Carbylamine
- Nitrous Acid Test
- Azo-Dye Test
Materials Required
- Aniline
- Potassium hydroxide
- Sodium nitrite solution
- Chloroform
- Dilute hydrochloric acid
- Concentrated hydrochloric acid
- Sodium hydroxide solution
- Benzene sulfonyl chloride
- β-Naphthol
- Test tubes
- Test tube holder
- Bunsen burner
Solubility Test
Amines are the organic compounds that have appreciable basicity and so they dissolve in mineral acids like hydrochloric acid.

Procedure
- Take the given organic compound to be tested in a clean test tube.
- Add 2 to 3 drops of concentrated hydrochloric acid.
- Shake well the solution and observe the solubility.
Note: If it is soluble in mineral acid then it may be an amine.
Litmus Test
Amines have appreciable basicity and turn red litmus blue.Amines are basic in nature.This test is also not a confirmation test for amines.

Procedure
- Take the moist red litmus paper.
- Place one to two drops of given organic compound on it.
- Observe the change in the colour of red litmus paper.
Note: The change in the colour of red litmus paper shows the given organic compound is a base. (may be amine)
Carbylamine Test
This test is also called isocyanide test, because when amines are treated with chloroform in the presence of alkali, isocyanide is formed. Both aliphatic and aromatic amines gives positive for this test. Secondary and tertiary amines do not give this test.
Procedure
- Take potassium hydroxide solid in a clean test tube and add 2ml of ethanol to it.
- Warm the test tube until the potassium hydroxide dissolves completely in it.
- To this add a few drops of chloroform and shake well.
- To the above mixture add a small amount of given organic compound to be tested and warm gently.
- Observe the odor of the resulting mixture.

Note: An unpleasant odor confirms the presence of primary amine.
Caution
1. Carpytomine (isocyanide) is highly poisonous so do not inhale its vapours.
2. Destroy carbylamine with cdnc. HCl after the test and then throw into sink.
Nitrous Acid Test
This test can be used to identify primary, secondary and tertiary amines. An aromatic primary amine reacts with nitrous acid forms a diazonium salt and at higher temperatures the formed diazonium compounds undergoes decomposition.

Procedure
- Dissolvethe given organic compound in concentrated hydrochloric acid and water in a clean test tube.
- Cool the solution in ice bath for 0-5oC.
- To the cooled solution add sodium nitrite dropwise and mix well.
- Test the solution for the presence of free nitrous acid.
- Add excess of nitrous acid solution if there is an absence of nitrous acid.
Note: Appearance of bubbles shows the presence of primary amines, yellow oily liquid shows the presence of secondary amines and formation of nitrite salts conforms the presence of tertiary amines.
Azo-Dye Test
This test is performed in order to confirm the presence of aromatic amines. Aromatic amines forms a diazonium salts when treated with HNO2. These diazo compounds couple with phenols like β-naphthols forms an orange azo dye which is insoluble in water.


Procedure
- Simultaneously take three test tubes and mark it as A, B and C.
- In test tube A dissolve the given organic compound in 2ml of dilute hydrochloric acid.
- In test tube B prepare aqueous solution of sodium nitrite solution.
- In test tube C dissolve naphthol in dilute sodium hydroxide.
- Place all the three test tubes in an ice bath.Now add the contents in test tube B to the mixture in test tube A.
- The resulting mixture is added to the contents in test tube C.
- Stir the mixture with a glass rod and observe the changes in the test tube.
Note: The presence of red orange dye conforms the presence of aromatic amines.
Distingushing Test for Primary , Secondary and Tertiary Amines
Hinsberg Test
The reagent used in this test is benzene sulfonyl chloride. Amines reacts with benzene sulfonyl chloride in the alkaline medium.
Procedure
- Take the given organic compound to be tested in a clean test tube.
- Add 2ml of sodium hydroxide solution and 1ml of benzene sulfonyl chloride to it.
- Shake the mixture well and add water to dilute the solution.
- Cool the mixture under tap water and observe the formation of precipitate.
- If precipitate formed then treat it with concentrated hydrochloric acid.


Results
- Tertiary amine. Precipitate in the test-tube. It dissolves in cone. HCl.
- Secondary amine. Precipitate in the test-tube. It does not dissolve in cone. HCl.
- Primary amine. No precipitate (clear solution). On addition of cone. HCl, insoluble material is seen.